Health Canada issued recalls for a number of products including deodorant, injectable peptides, eye drops and supplements.
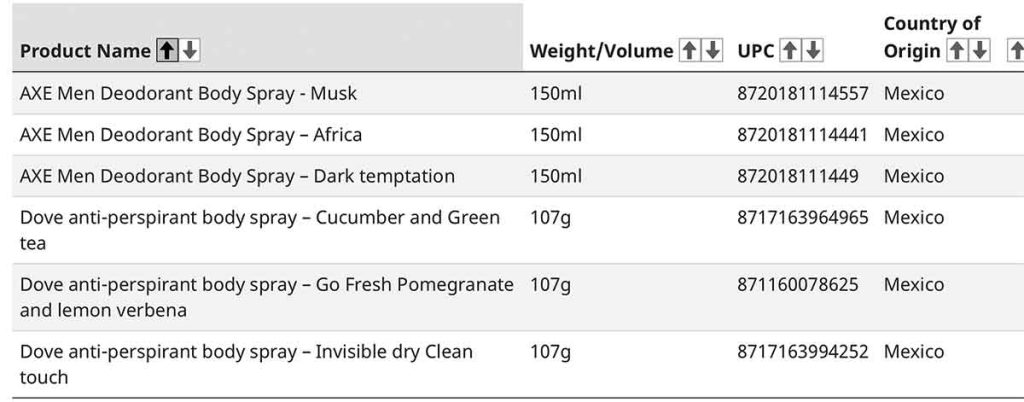
The health agency issued a recall on April 10 for Axe men’s deodorant body spray in various scents and Dove anti-perspirant in various scents due to a lack of hazard labels.
“These products do not meet the Cosmetic Regulations in Canada. Specifically, the products do not have mandatory hazard labelling,” states the recall. “The products are missing required hazard symbols, signal words on the front of the containers and safety information. Improper labelling could result in misuse of the product and lead to serious injury.”
The products are distributed by TJ Trading Co. Inc. The company reported that 384 units of the affected products were sold in Canada.
The products were sold from January 2026 to March 2026.
As of April 1, 2026, the company received no reports of incidents in Canada, and no reports of injury, states the recall.
The health agency issued a warning on April 9 advising consumers to “think twice before injecting peptides bought online” as unauthorized products can cause serious health risks.
Unauthorized injectable peptides are sold and marketed as anti-aging, bodybuilding and wellness products as well as for athletic performance, injury recovery, sleep and mental focus.
“Do not buy or use these products. If you have used them and feel unwell or have concerns, talk to a healthcare professional right away. Only buy prescription drugs from licensed pharmacies,” states the warning. “Check labels: authorized drugs for sale in Canada have an 8-digit Drug Identification Number (DIN) printed on the label.”
The unauthorized products include BPC-157, CJC-1295, DSIP, Epitalon, GHK-Cu, HCG, Ipamorelin, KPV, Melanotan I and II, MOTS-C, NAD+, SS-31, TB-500 and Retatrutide.
“In Canada, peptides are generally regulated as prescription drugs. Authorized peptide drugs should only be used under the care of a licensed healthcare professional, such as a doctor, to treat specific conditions,” states the warning. “Health Canada reminds consumers that decisions about prescription medication should be made in consultation with a healthcare professional and should only be purchased from a licensed pharmacy.”
Three brands of Jamieson Laboratories supplements were recalled in a notice issued April 1 because tamper-evident seals were defective.
The products are Joint Pain Relief Capsules, Magnesium Bisglycinate Capsules and Maca (Lepidium meyenii) Capsules.
And nine ophthalmic solutions from Labtician Ophthalmics Inc./Aved brand were recalled in a notice issued April 2.
None of the recalled products had obtained market authorization.
Visit Health Canada’s Recalls and Safety Alerts website for full details, including lot numbers, of all the above affected products.
